NiaHealth Research Process

Introduction
NiaHealth aims to empower people to extend and enrich their functional lifespan (i.e. their healthspan) through early identification and management of risk factors that contribute to the development of major causes of morbidity and mortality, including cardiovascular disease, metabolic dysfunction, neurodegenerative disease, and cancer.
To achieve this, NiaHealth offers a personalized health assessment that takes into account demographics, medical history, physiologic measures, biomarkers, and data from wearable devices. Based on an individual's identified health priorities, NiaHealth provides tailored recommendations for optimizing healthspan, with the opportunity for deeper insight from a one-on-one consultation with a clinician.
Diet and lifestyle strategies are foundational to the management recommendations provided by NiaHealth. In the event that additional diagnostic testing or pharmacologic therapy are indicated based on the outcome of a NiaHealth assessment, clients are provided with education and correspondence for their primary care provider to facilitate shared decision-making. NiaHealth also offers clients the opportunity to track their progress with serial assessment.
What sets NiaHealth apart from other providers in the preventative medicine and longevity space is a commitment to evidence-guided practice. All steps of the NiaHealth process–from determining which tests to incorporate into health assessments, to lifestyle recommendations provided to clients and referrals to primary care–are informed by clinical research evidence. This document describes the process used by the NiaHealth Research Team to evaluate existing evidence and how this evidence is implemented into our platform.
NiaHealth approach to evidence-guided practice
Decisions about whether to incorporate a test or management recommendation into the NiaHealth platform are made based on studies in humans that have demonstrated efficacy, safety, and applicability to NiaHealth users. Where possible, recommendations are based on clinical practice guidelines, in conjunction with well-conducted systematic reviews and/or meta-analyses (e.g. Cochrane Systematic Reviews), randomized clinical trials (RCTs preferred for management strategies), observational cohort studies (particularly from well-established, well-described cohorts such as CHMS and NHANES), cross-sectional studies, or expert consensus. We do not recommend investigations or management based on case reports, case series, or preclinical studies (i.e. animal studies or translational studies).
Where evaluation measures are being assessed, observational studies (and systematic reviews/meta-analyses of observational studies) often provide the most applicable evidence. However, where management strategies (interventions) are being assessed, RCTs (and systematic reviews/meta-analyses of RCTs) are preferred.
While the majority of the NiaHealth evidence synthesis and implementation approach is in line with the accepted definition of evidence-based practice–i.e. based on available evidence (ideally systematic reviews with meta-analyses), clinical experience, the values and preferences of patients, and reported using transparent standards (Dhurandhar et al., 2025; Ghosh et al., 2024)–some tests in the platform do not have enough evidence to justify widespread use in clinical practice. For this reason, while all included tests have been evaluated in human studies, we consider our approach evidence-guided rather than evidence-based.
NiaHealth research process
The NiaHealth research process adapts established methodology for conducting scoping reviews, systematic reviews, and meta-analyses with the view of balancing quality and efficiency. The process is shown in the flow diagram below, followed by a detailed description of each step.
NiaHealth research process flow
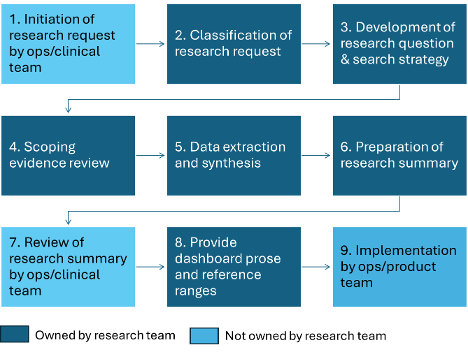
1. Initiation of research request - ops/clinical team
Requests are initiated by the Clinical Director, Chief Operating Officer, or Chief Executive Officer. Prioritization of requests is done by the requestors.
2. Classification of research request
Research requests typically involve either assessment of a test (for screening, diagnosis, or risk classification) or an intervention (for prevention or management). The research team classifies request as follows:
- Test
- Test type: survey, physiological measure, biomarker, other risk assessment tool
- Reason for research: recommendation on whether to bring new test into NiaHealth platform (e.g. should NiaHealth offer a multi-cancer early detection test to clients?), optimization of delivery of an existing test (e.g. are current NiaHealth testosterone reference ranges consistent with the evidence?)
- Intervention
- Intervention type: diet or lifestyle, supplement, medication, other
- Reason for research: review of evidence to support a specific intervention in a certain condition (e.g. is there evidence that a client would benefit from taking probiotics during an upcoming course of antibiotics?), summary of all evidence-based management recommendations for a certain situation (e.g. if a client has a low Omega 3 Index, what recommendations are likely to improve this measure?)
3. Development of research question & search strategy
Research question
The PICO framework is used to develop research questions, with modifications as needed depending on the type of research request (Hosseini et al., 2024). The PICO approach involves identifying the relevant Patient/Population, Intervention (or Exposure/Risk/Result) of interest, Comparator/Control, and Outcomes. This information is then utilized to inform the literature search strategy with the goal of finding and evaluating the available evidence.
P = Patient or population
I = Intervention
C = Comparison/control
O = Outcome
The PICO approach is focused on interventional studies and will be adapted as follows for other types of research requests:
Test / Diagnostic accuracy (PIRD): Population, Index test, Reference test (Gold standard), Diagnosis of interest
Test / Disease risk factor (PEO): Population, Exposure, Outcome
Intervention (PICO): Patient/Population, Intervention, Comparator, Outcome
Some examples of research questions developed using the PICO approach are shown below:
Test (diagnostic accuracy)
Among the general adult population (Population), how does a ctDNA-based blood test for colon cancer (Index test) compare with current guideline-based screening recommendations (Reference test) in diagnosing cancer (Diagnosis of interest)?
Test (disease risk factor or cause)
Among the general adult population (Population), is there a relationship between daily step count (Exposure) and mortality (Outcome)?
Intervention
Among people with low Akkermansia levels (Population), does treatment with an Akkermansia-containing probiotic (Intervention) compared to placebo (Comparator) result in weight loss (Outcome)?
Search strategy
Using terms from the research question, a search strategy is developed according to the following template:
("<test name>" OR "<analyte>" OR "<common synonyms>") AND ("diagnostic accuracy" OR sensitivity OR specificity OR "likelihood ratio" OR AUC OR "prognostic" OR "risk stratification" OR "clinical utility") AND (adult OR adults) NOT (animals)
4. Scoping evidence review
Using the research question and search strategy developed in step 3, an initial literature search is undertaken. In most cases, NiaHealth uses a scoping review strategy for this, although a more specific systematic review search strategy may be used for narrow research questions. In either case, the literature search incorporates medical databases, grey literature, and an artificial intelligence (AI)-assisted review.
Medical databases
Medical databases are the primary source of high-quality research that NiaHealth relies upon for decision-making. The search strategy is applied to one or both of the following medical databases:
- PubMed/Embase
- OVID
Depending on the nature of the research question, the search may be limited to:
- Only studies in humans
- Only reviews
- Only systematic reviews and/or meta-analyses
- Only RCTs
Depending on the research request, searches of the following databases may also be conducted: Cochrane, Web of Science/Scopus.
Grey literature
Grey literature has not been published through traditional means and is therefore often excluded from large databases and other mainstream sources as it does not contain the necessary bibliographic information. Inclusion of grey literature in a research review can reduce bias, improve thoroughness, and find the most up-to-date evidence.
Therefore, every research request will involve a review of the grey literature. At minimum, this includes:
- A search of the topic in Google with review of the first 5-10 pages for relevant authorities, organizations, or stakeholders, and websites that may include relevant grey literature.
- Review of any relevant professional society guidelines, healthcare technology assessments (e.g. from CADTH or NICE)
In situations where traditional literature is sparse, a more detailed grey literature review may be undertaken, including review of additional grey literature resources (here).
Artificial Intelligence (AI)
AI tools such as ChatGPT can provide a rudimentary overview of existing research (Fabiano et al., 2024). ChatGPT is incorporated into the NiaHealth literature search process, as an adjunct to medical database and grey literature searches. As LLMs such as ChatGPT are known to hallucinate and to provide incorrect references, all claims and references will be cross-checked by the research team to verify accuracy.
NiaHealth ChatGPT Research Best Practices
AI-assisted searching is done using ChatGPT, in Deep Research mode.
Identification of key literature
Following a search of medical databases, grey literature, and an additional GPT-assisted search, the research team reviews all identified papers at a high level (e.g. titles and abstracts) and identifies key literature. This process is similar to the title and abstract review in a systematic review.
According to the evidence hierarchy, systematic reviews and meta-analyses are prioritized, followed by well-designed studies that are appropriate for the research question. When the research question focuses on a test and/or disease risk factor, results of observational studies carried out in well-described, population-based cohorts are prioritized. A list of such cohorts is provided in Observational Cohorts Used in NiaHealth Research. When the research question focuses on an intervention, RCTs are prioritized.
The reference lists of relevant papers (particularly narrative reviews, systematic reviews, and meta-analyses) will be reviewed for potential key literature that was not identified in the initial literature search.
5. Data extraction and synthesis
Key literature identified in step 4 is reviewed in detail by the research team and relevant data extracted. This is similar to the full text review process in a systematic review.
Data extraction
The following data is extracted from identified key literature:
- Study meta-data: design, setting, sample size, demographics, reference standard (for tests), comparator (for clinical trials)
- Results/outcomes: test performance, effects on management/outcomes, adverse events
- Interpretation: focusing on relevance and applicability to NiaHealth
The data extraction process may also include a Risk of Bias assessment, using the Cochrane Risk of Bias 2 tool for interventional studies or the Newcastle-Ottawa Scale (NOS) for observational studies. Depending on study design, the following tools may also be utilized: QUADAS-2 (diagnostic), ROBINS-I (observational), AMSTAR 2 (systematic reviews).
Data synthesis
The evidence synthesis includes a holistic assessment of data extracted from key literature in order to answer the research question(s). Typical questions addressed in the synthesis of research reviews for tests (new tests or optimizing existing tests) and interventions are described below.
Review of a new test
Evaluation of potential new tests is guided by the recommendations of the Agency for Healthcare Research and Quality (Matchar, 2012; St John et al., 2021). Specifically, the following questions are asked:
- Are the test characteristics acceptable?
- Can the test result be linked to an improved health outcome?
- Can the test inform patient treatment decisions beyond standard (or existing) measures?
- How many patients must be screened with the test in order to identify one with the disease (or risk factor) or condition?
- Are there potential harms associated with false positive test results?
- Do the benefits justify the incremental costs of testing if large numbers of patients are screened to identify a small number of potential cases with the disease?
- Are there established clinical decision limits (CDLs)? If not, does evidence exist to guide the internal development of CDLs? If not, is it possible to set population/normative-based reference intervals? (More information provided in Determining Reference Intervals and Clinical Decision Limits)
- Is there an appropriate reference change value that should be used for interpreting serial tests? (More information provided in Determining Reference Change Values)
- What is the quality of the evidence?
- What is the strength of the evidence?
Questions specific to NiaHealth:
- Is the current evidence applicable to the NiaHealth population?
- Are results actionable to NiaHealth clients? Are actions likely to impact health outcomes?
- Is there a feasible management/follow-up plan for NiaHealth clients if test results are abnormal?
Optimization of an existing test
Questions around optimization of a test currently being offered by NiaHealth may involve some of the same questions listed above, although typically they will be targeted specifically to certain questions (e.g. test characteristics, disease associations, recommended reference ranges)
Review of an intervention
Evaluation of interventions focuses on efficacy, safety, and generalizability. The following questions are used for guidance:
- Are there high-quality systematic reviews, meta-analyses, or RCTs to support the intervention?
- If not, what is the highest quality of evidence available?
- Does the existing evidence support the intervention?
- Is the intervention effective?
- What is the relative risk reduction, absolute risk reduction, and number needed to treat?
- What health outcomes does to intervention impact?
- Is the intervention safe? What adverse effects have been reported?
Questions specific to NiaHealth:
- Is the current evidence applicable to the NiaHealth population?
- Is the intervention actionable for NiaHealth clients? Are actions likely to impact health outcomes?
6. Grading of evidence
The research team uses the GRADE scoring system (reviewed in Principles of Evidence-Based Practice) to determine the strength of the evidence (Prasad, 2024) according to the following scale:
- High certainty: evidence is robust enough to draw a conclusion
- Moderate certainty: evidence supports a conclusion but further research may impact confidence in the estimate
- Low certainty: additional research is likely to have an important impact on the confidence in the estimate OR available evidence is insufficient to support any firm conclusions
The GRADE handbook describes four categories (high, moderate, low, very low), with the note that the low and very low categories may be combined if appropriate. In the NiaHealth setting, we have chosen to combine the low and very low categories.
7. Preparation of Research Summary
Based on the results of the evidence synthesis, the research team prepares a report according to the NiaHealth Research Summary Template, containing the following sections (these may vary depending on the nature of the research request):
For research questions regarding tests:
- Title
- Summary
- Description
- Rationale for Test
- Testing Considerations
- Results
- Clinical decision limits
- Conventional reference ranges
- Interpretation
- Reference change values
- Management
- Quality and certainty of evidence
- Applicability to NiaHealth
- Recommendations
- Key Paper Summary
- References
For research questions regarding interventions:
- Title
- Summary
- Description
- Rationale for intervention
- Safety
- Clinical use
- Quality and certainty of evidence
- Applicability to NiaHealth
- Recommendations
- Key Paper Summary
- References
8. Review of research summary - ops/clinical team
Following completion of the research summary, which includes evidence-guided recommendations from the research team, the operations and clinical teams decide whether to action the research recommendations and/or whether further research is required before reaching a decision.
9. Provide dashboard prose and reference ranges
For any changes being made to the NiaHealth product as a result of the research review, the research team provides dashboard prose, reference ranges, and other information required by the operations and product teams to implement the changes. The NiaHealth Research Implementation Template is used for this.
10. Implementation - ops/product team
Changes are implemented by the operations and product team. Information provided by the research team for implementation (e.g. dashboard prose, reference ranges) will be added to the test spec by the product team.
Our editorial standards & process
At NiaHealth, our mission is to make proactive health possible for all Canadians—by combining science with humanity. We believe that rigorous, evidence-informed health information should never feel out of reach. Every word we publish is intentional. We choose language that empowers rather than overwhelms, clarifies rather than complicates, and respects the lived experiences behind every health question. Learn more here.
